There are many methods for determining protein concentration. The Bradford assay (Coomassie Brilliant Blue method) is one of them, utilizing colorimetric detection for simple and easy operation.

Experimental Principle:
The Bradford protein assay is based on the principle of protein-dye binding. It is a rapid and sensitive method for the quantitative determination of trace protein concentrations.
Coomassie Brilliant Blue R-250 dye binds with proteins in an acidic solution, causing the maximum absorption peak ($\lambda_{max}$) to shift from 465 nm to 595 nm. The color of the solution changes from brownish-green/bluish-black to blue. By measuring the increase in light absorption at 595 nm, the amount of protein bound to the dye can be determined. Research has found that the dye primarily binds to basic amino acids (especially arginine) and aromatic amino acid residues in proteins.

Dye does not easily adhere to clean dishes.
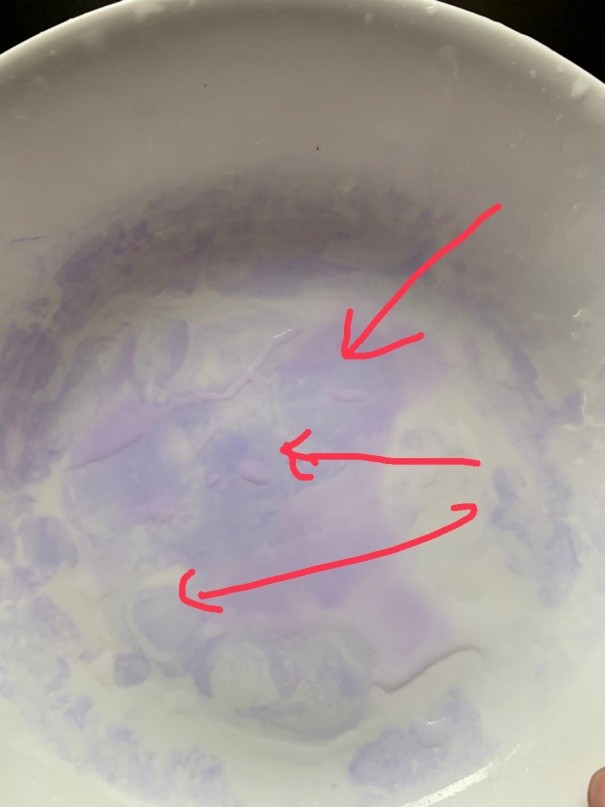
Significant blue color indicates protein residue.
Outstanding Advantages of This Method
(1) Fast, simple, and highly sensitive. It is estimated to be approximately four times more sensitive than the Lowry method, with a minimum protein detection limit of 1 mg. This is because the color change after the protein binds with the dye is significant, and the protein-dye complex has a higher extinction coefficient.
(2) Easy to operate, requiring only one reagent. Measuring a single sample takes only about 5 minutes. Since the binding process takes only about 2 minutes and the color remains stable for up to an hour (with peak stability between 5 to 20 minutes), it does not require the strict time control of the Lowry method.
(3) Few interfering substances. Common substances like $K^{+}$, $Na^{+}$, $Mg^{2+}$ ions, Tris buffer, sugars, glycerol, mercaptoethanol, and EDTA do not interfere with this assay.
Disadvantages of This Method
(1) Due to varying levels of arginine and aromatic amino acids in different proteins, there can be significant deviation when measuring different proteins. To reduce this, $\gamma$-globulin is typically used as a standard protein for calibration curves.
(2) Some substances still interfere with the measurement, primarily detergents such as Triton X-100 and Sodium Dodecyl Sulfate (SDS).
Testing Method:
1. Reagent Preparation:
Bradford Reagent: Dissolve 10 mg of Coomassie Brilliant Blue R-250 in 5 mL of 95% ethanol, add 10 mL of 85% phosphoric acid, and dilute with distilled water to 100 mL.
2. Spray the Bradford reagent evenly onto the dish. For any areas not reached, rotate the dish to ensure the reagent is distributed across the entire surface.
3. Observe the color of the reagent on the dish. A significant blue color indicates the presence of protein residue.

Dye does not easily adhere to clean dishes.

Significant blue color indicates protein residue.
Precautions
(1) Read the results within 5-20 minutes after adding the reagent, as the color is most stable during this window.
(2) During testing, a small amount of the protein-dye complex may adhere to the surface of the dish. After testing, this blue residue can be cleaned off using ethanol.